
Again, the pentagonal bipyramidal structure is more stable. When we proceed in a similar manner for sp $^3$d $^3$, we have two options: heptagonal planar (bond angle $51.43°$) and pentagonal bipyramidal (bond angles $72°$ and $90°$. Thus the hybridization of XeF2 molecule is sp3d. Hence the hybridization of the central atom Xe is sp3d. The arrangement of the electrons of Xenon changes to s2 p5 d1 with two unpaired electrons. Of the two, trigonal bipyramidal is obviously a more stable structure (bond angles $90°$ and $120°$) than pentagonal planar (bond angle $72°$) ( $3$D here). sp3d Whereas in XeF2, the Xe molecule has an excited state. The instant we do that, we are left with only two options: trigonal bipyramidal and pentagonal planar.
Therefore, for symmetry about the plane, we must keep a third bond on the plane. We cannot put $2$ bonds on one side and $1$ bond on the other side, as that would be unsymmetrical ( $3$D here). Using the condition above, we have to arrange $3$ bonds around the plane, such that they are symmetric about it. It has a 3s and three 3p orbitals, so it should use one of its 3d orbitals to type the fifth bond.These orbitals are hybridized to type 5 sp3d orbitals and thus type a trigonal bipyramid.

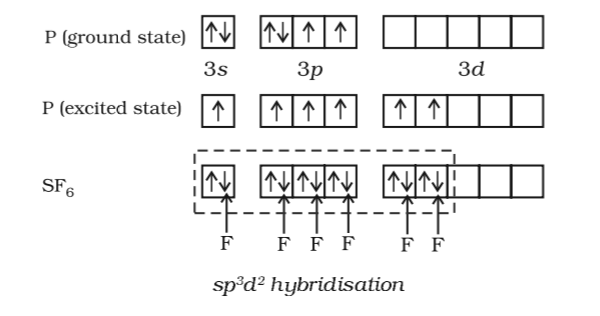
Can you explain this answer EduRev JEE Question is disucussed on EduRev Study Group by 113 JEE Students. The P atom wants 5 orbitals to type the 5 P-Cl bonds. Obviously, the two bonds form a plane ( $3$D here): Dec 30,2021 - In allene (C3H4), the type(s) of hybridization of the carbon atoms is (are):a)only sp2b)sp2 and spc)sp and sp3d)sp2 and sp3Correct answer is option B. In the first step, one electron jumps from the 2s to the 2p orbital. Now, let’s see how that happens by looking at methane as an example. I'm going to show that there are no better structures satisfying this condition for sp $^3$d and sp $^3$d $^3$ hybridisation.įor sp $^3$d, we're going to first arrange any two bonds at some angle (unknown). Hybridization is a theory that is used to explain certain molecular geometries that would have not been possible otherwise. Note that this condition is satisfied by trigonal planar, tetrahedral, trigonal bipyramidal, octahedral and pentagonal bipyramidal geometry. Why are these arrangements preferred in favor of one with a simple symmetric arrangement of orbitals (which, I think, would minimize the repulsion)?įor a structure to be perfectly symmetric, it must satisfy this condition:įor any two bonds, the plane passing through them must divide the molecule into two symmetric halves. A molecule in which the central atom is $sp^3d^2$ hybridized ($\ce$) with five orbitals crammed into the equatorial plane and two sticking out above and below it.
